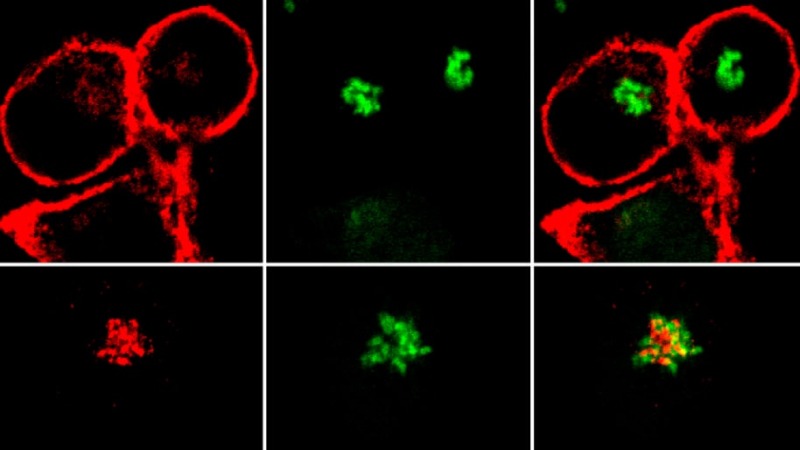
Investigators: David Sherman, Ph.D., and Kathleen Collins, M.D., Ph.D.
Disease problem: Approximately 39 million people wordlwide are living with human immunodeficiency virus (HIV). While treatments are available to keep the virus under control, there is no cure for the virus. The virus has the ability hide away in remote, long-lived human cells, where it can escape the total eradication from the body's natural immune system and antiviral medications.
Partnership: The Innovation Partnership supported a collaboration between Sherman, an LSI faculty member, and Collins, a microbiologist at the U-M Medical School, to develop a novel approach to fighting HIV.
Over the past decade, Collins and her team have shown how Nef, a protein made by HIV, is a key player in the virus’s ability to hide from the defenders in the body’s immune system. Collins figured that if she could find a drug that could stop Nef, she and her team could remove HIV’s camouflage and allow the body to fight it off as it would a cold virus. They tested a huge library of potential drugs, but none worked.
Then, the U-M Center for Chemical Genomics, based at the U-M Life Sciences Institute—where Sherman’s lab is located—helped Collins test more than 26,000 microorganism-derived extracts from a library built primarily from Sherman’s expeditions. After narrowing the results down to 11 promising compounds, they zeroed in on the three with the most potential to inhibit Nef. All were made by marine actinomycetes bacteria. Now, Collins and Sherman are collaborating to develop the results further.
“We’re still at the early stages of this project,” Collins says. “No one has developed a Nef inhibitor with the level of activity that we’re seeing. If this works, we could essentially harness a person’s own immune system to wipe HIV out of the body.”
Learn More
"The Quest for a True AIDS Cure — in a Coral Reef," Michigan Health Lab Blog